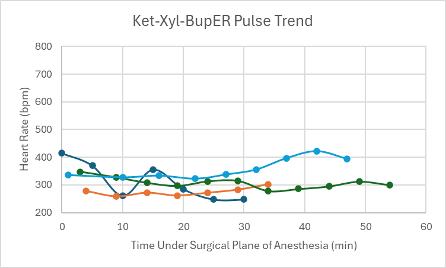
Figure 1: Heart rate trend in mice under a surgical plane of anesthesia after the administration of the Ket-Xyl-BupER cocktail.

Medicine
Surgical Anesthetic Potential of Injectable Cocktails Containing Acepromazine, Alfaxalone, Dexmedetomidine, Ketamine, Midazolam, Telazol, and Xylazine with and without the Addition of BuprenorphineER in Mice
Spring 2025, Volume 23, Issue 2
Eleanor Harrison ’26, and Dana LeMoine*
Abstract
Common anesthetics in the field of veterinary medicine have many well-known imperfections. These include popular choices, such as inhalant anesthetics and injectable xylazine and ketamine cocktails. However, less popular, alternative injectable anesthetic cocktails do not have as much data available concerning their effectiveness in reaching a surgical plane of anesthesia in laboratory mice. Therefore, during this study, the surgical anesthetic potential of four experimental injectable anesthetic cocktails and two control cocktails were observed in transgenic C57BL6, transgenic DBA, CD-1, Heterozygous Nude Nu/J, and Agouti mice. The following cocktails used for this study were: Ket-Xyl + BupER, Ket-Dex +BupER, Tel-Dex +/- BupER, Ket-Dex-Ace, Alfa-Dex +/- BupER, and Alfa-Mid +/- BupER. The cocktails with the most consistent results and most desirable trends in heart rate, SpO2, and respiratory rate for the duration of the surgical plane of anesthesia were Ket-Dex +BupER, Ket-Dex-Ace, and Alfa-Dex +BupER. However, Ket-Dex-Ace, had a very long duration of sedation even after reflexes were gained, so it is recommended for longer procedures/surgeries. Ket-Xyl +BupER showed inconsistency in the mice reaching a surgical plane of anesthesia, Tel-Dex showed variable and dangerous vitals throughout the surgical plane of anesthesia, and Alfa-Mid +BupER showed variable vital trends and inconsistent lengths of a surgical plane of anesthesia. Thus, these cocktails are not recommended.
Introduction
Anesthetics are essential to laboratory animal research to allow for the completion of procedures and surgeries. Inhalant anesthetics are preferred since it is easier to adjust the amount of anesthetic being administered, and the plane of anesthesia reached is generally more consistent. However, in the field of laboratory animal research, where mice are the primary research model, inhalant anesthetics are not ideal for some laboratory procedures and surgeries1. Popular alternatives for inducing anesthesia in mice include injectable anesthetic cocktail combinations of ketamine with xylazine or dexmedetomidine. Unfortunately, these combinations are known to be unreliable and cause undesirable side effects such as cardiac depression, bradycardia, and a 40% increase in death rate2.
Another concern that has become apparent in recent years is the introduction of xylazine and ketamine into the illicit drug supply chain within the United States3. Due to these developments, heavier restrictions have been placed on these drugs, causing their supply to decrease and making them more difficult to obtain for use in the veterinary field.
In veterinary medicine, anesthetic cocktails generally include a dissociative agent, such as ketamine, and an α2-adrenergic agonist, such as xylazine or dexmedetomidine1. Dissociative agents are known to provide muscle relaxation, sedation, and anesthesia, while α2-adrenergic agonists provide sedation and analgesia4,5. Though ketamine is a popular choice, there are some alternatives to this drug that show potential. One possible substitute is Telazol since it contains a drug called tiletamine that acts as a muscle relaxer and can produce an anesthetic state6. Another drug that could replace ketamine is alfaxalone, a neuroactive steroid that acts as a GABA agonist and is a sedative that results in anesthesia and muscle relaxation7,8. There has been some research that shows potential in combining alfaxalone with xylazine to create an effective anesthetic cocktail7,8. However, this combination has shown high mortality rates in mice and other undesirable effects7, so this study decided to look into other drug combinations using alfaxalone without xylazine.
Xylazine and dexmedetomidine are both α2-adrenergic agonists, however, dexmedetomidine has a greater specificity for the α-2 receptor4. For this reason, this study chose to evaluate cocktails that use dexmedetomidine instead of xylazine to see if they show similar potential to cocktails that contain xylazine. Some of the cocktails evaluated also included midazolam or acepromazine. Midazolam is a sedative, and is a useful coinduction anesthetic agent to alfaxalone4,9, while acepromazine is a sedative that acts through postsynaptic dopaminergic D2 receptor antagonism9. Some research has been done that shows potential in using these drugs as anesthetic agents in other species9, so this study decided to evaluate their effectiveness in cocktail combinations for mice.
Past research has also shown that the addition of Buprenorphine ER, a synthetic opioid that produces analgesia4, can help create a shorter induction period and a longer, more stable anesthetic state4,8. Furthermore, BupER is routinely used to provide postoperative analgesia in animals, so it is commonly combined with anesthetics before a procedure or surgery. Thus, some of the experimental cocktails used in this study also included the administration of Buprenorphine ER to replicate routine conditions and evaluate the effectiveness it had in helping produce a surgical plane of anesthesia.
The experimental cocktails tested were Telazol with Dexmedetomidine, with or without Buprenorphine ER (Tel-Dex, Tel-Dex-BupER), Ketamine with Dexmedetomidine and Acepromazine (Ket-Dex-Ace), Alfaxalone with Dexmedetomidine, with and without Buprenorphine ER (Alfa-Dex, Alfa-Dex-BupER), and lastly Alfaxalone with Midazolam, with or without Buprenorphine ER (Alfa-Mid, Alfa-Mid-BupER). The control cocktails we used throughout the study are considered the standards for injectable anesthetics and included Ketamine with Xylazine with the addition of Buprenorphine ER (Control A), and Ketamine with Dexmedetomidine with the addition of Buprenorphine ER (Control B). The purpose of this study was to evaluate if experimental cocktails provided a surgical plane of anesthesia for at least 20 minutes, similar to the control cocktails.
Materials and Methods
Animal Information
Experiments were done on adult mice between the ages of 8 and 40 weeks old. A variety of males and females were used. Different strains of female mice included Heterozygous Nude Nu/J (n = 6) and transgenic C57BL6 background (n = 19); specific transgenic C57BL6 backgrounds included female mice of CasQI background (n = 3), TNF background (n = 3), Y5225 x G100S/Orai1 background (n = 2), C57BL6-TKF (n = 1) and C57BL6-Y5225 background (n = 1). Female weights ranged from 18g to 33g. Different strains of male mice included Agouti (n = 3), transgenic DBA CD47 +/- (n = 2), CD-1 (n = 3), and C57BL6 background (n = 18); specific transgenic C57BL6 backgrounds included males of CasQI background (n = 2), TNF background (n = 1), TGFBpg-mTmG-TMF background (n = 1), GFBP7-mTmGITMF background (n = 1), TMFR2-KO background (n = 2), and KPC background (n = 4). Male weights ranged from 18g to 57g. All mice were housed in cages with Biofresh ⅛” Performance Plus bedding and were fed irradiated rodent chow food with HYDROPAC ® water. Housing conditions were 64-79℉ and 30-70% humidity10.
All mice used in this study were free of lymphocytic choriomeningitis, mycoplasma pulmonis, mouse adenovirus, mouse cytomegalovirus, rodent coronavirus, sialodacryoadenitis virus, respiratory syncytial virus, myobia musculi, radfordia affinis, radfordia ensifera, myocoptes musulinus, murine norovirus, mouse parvoviruses, ectromelia, monkeypox, rabbitpox, mouse rotavirus, syphacia obvelata, syphacia muris(rat), aspiculuris tetraptera, pneumonia virus of mice, reovirus types 1, 2 and 3, sendai virus, Theiler’s murine encephalomyelitis virus, and rat theilovirus. Each mouse was only anesthetized once. All protocols for the experimental procedures were approved by UCAR through the University of Rochester School of Medicine and Dentistry.
Experimental Design
Four different anesthetic cocktails were experimented with throughout this study. The first cocktail evaluated was Tel-Dex administered intraperitoneally (IP) with and without the addition of BupER administered subcutaneously (SQ). The second cocktail was Ket-Dex-Ace administered IP. The third cocktail was Alfa-Dex administered SQ with and without the addition of BupER SQ Lastly, the fourth cocktail was Alfa-Mid administered SQ with and without the addition of BupER SQ. For all cocktails, a low dose was administered first and then adjusted accordingly in order to determine the dosage required to reach a surgical plane of anesthesia. For the Tel-Dex and Alfa-Mid cocktails, BupER was added if the cocktail alone failed to achieve a surgical plane of anesthesia for at least 20 minutes in the majority of mice. For the Alfa-Dex cocktail, BupER was administered in addition to the cocktail for half of the mice in the experimental group (n =6).
A surgical plane of anesthesia was determined through the loss of righting reflex (LORR) and loss of the pedal withdrawal reflex (PWR). To evaluate LORR, all mice were manually placed in dorsal recumbency. If the mouse was able to right itself by placing one or more paws back onto the surface they were lying on, they were determined to still have their righting reflex (RR). To evaluate the loss of the PWR, a reflex instrument (6.65 300g Touch Test ®) was used to apply a consistent noxious stimulus of 300 g. If any muscle twitch was observed from the application of noxious stimuli, the mouse was determined to still have its PWR. A surgical plane of anesthesia was defined as the duration for which a mouse did not have its PWR or any other spontaneous movement (i.e. blinking, tail twitching, head movement).
Drugs and Dosing
The anesthetic drugs that were used to create these cocktails were Ketamine (Ketamine HCl, Hikma, 100mg/mL), Xylazine (Rompun, Dechra, 100mg/mL), Dexmedetomidine (Dexmedesed, Dechra, 0.5mg/mL), Alfaxalone (Alfaxan ® Multidose, 10 mg/mL), Acepromazine (Acepromazine maleate, Vet One, 10mg/mL), Midazolam (Mfd. by Hikma, 5mg/mL), and Telazol (Zoletil, Virbac, 100mg/mL). To create the desired cocktail volumes, sterile saline solution (0.9% Sodium Chloride Injection, USP, by Hospira) was used. Some experimental groups also included the administration of Buprenorphine ER (Buprenorphine Base in Polymer Wedgewood Connect, 1 mg/mL) SQ in addition to the cocktail administration. The dosing range for the BupER was between 0.74-1.2 mg/kg.
| Cocktail Name | Cocktail Preparation | Low Doses | Mid Doses | High Doses |
| Tel-Dex | Telazol 17mg (0.17mL) Dexmedetomidine 0.21mg (0.42mL) Sterile Saline Solution (1.41mL) |
Telazol: 11 mg/kg Dexmedetomidine: 0.13-0.14 mg/kg |
||
| Ket-Dex-Ace Cocktail #1 | Ketamine 20mg (0.40mL) Dexmedetomidine 0.18mg (0.72mL) Acepromazine 0.40mg (0.08mL) Sterile Saline Solution (0.80mL) |
Ketamine: 50-55 mg/kg Dexmedetomidine: 0.45-0.49 mg/kg Acepromazine: 1.0-1.1 mg/kg |
Ketamine: 61 mg/kg Dexmedetomidine: 0.55 mg/kg Acepromazine: 1.2 mg/kg |
|
| Ket-Dex-Ace Cocktail #2 | Ketamine 15mg (0.30mL) Dexmedetomidine 0.20mg (0.80mL) Acepromazine 0.15mg (0.03mL) Sterile Saline Solution (0.87mL) |
Ketamine: 48-52 mg/kg Dexmedetomidine: 0.64-0.69 mg/kg Acepromazine: 0.48-0.52 mg/kg |
Ketamine: 67 mg/kg Dexmedetomidine: 0.89 mg/kg Acepromazine: 0.67 mg/kg |
|
| Alfa-Dex | Alfaxalone 5.0mg (2.0mL) Dexmedetomidine 0.05mg (0.4mL) Sterile Saline Solution (1.6mL) |
Alfaxalone: 50-52 mg/kg Dexmedetomidine: 0.50-0.52 mg/kg |
Alfaxalone: 76 mg/kg Dexmedetomidine: 0.76 mg/kg |
|
| Alfa-Mid | Alfaxalone 5.0mg (1.0mL) Midazolam 0.5mg (0.2mL) Sterile Saline Solution (0.80mL) |
Alfaxalone: 39-41 mg/kg Midazolam: 3.9-4.1 mg/kg |
Alfaxalone: 50 mg/kg Midazolam: 5.0 mg/kg |
Alfaxalone: 59-61 mg/kg Midazolam: 5.9-6.1 mg/kg |
| Control A | Ketamine 20mg (0.20mL) Xylazine 2.0mg (0.02mL Sterile Saline Solution (0.78mL) |
Ketamine: 79-84 mg/kg Xylazine: 7.9-8.4 mg/kg |
||
| Control B | Ketamine 15mg (0.15mL) Dexmedetomidine 0.20mg (0.40mL) Sterile Saline Solution (0.45mL) |
Ketamine: 47-52 mg/kg Dexmedetomidine: 0.63-0.69 mg/kg |
Ketamine: 65 mg/kg Dexmedetomidine: 0.87 mg/kg |
Table 1. Provides information regarding cocktail preparation and the doses that were administered to the mice throughout experimentation.
Anesthetic Depth and Physiologic Monitoring
To begin the calculations for dosing, the mice were weighed on a gram scale (Model CS 5000, Capacity 5000g x 1g, Ohaus Corporation, USA). Once a weight was taken, each mouse was injected with a designated anesthetic cocktail, and then monitored for their LORR. Once a LORR was reached the mice were placed onto an automated heating pad (ALA Scientific Instruments, HOT-1 Temperature Controller, Boatman 12V/14W) or a microwavable heating pad (Microwavable Warmer by K&H™ Pet Products) and eye lube (Neomycin and Polymyxin B Sulfates and Bacitracin Zinc Ophthalmic Ointment, USP by Bausch & Lomb Americas Inc.) was applied. The mice were then monitored for the loss of their PWR. The time when they lost their PWR was defined as T = 0. While under a surgical plane of anesthesia, the mice were continuously monitored every 5 minutes for their temperature, heart rate, peripheral oxygen saturation of hemoglobin (SpO2), respiratory rate, mucus membrane color, and PWR. Temperature was measured with a rectal probe (PhysioSuite Non-Invasive Monitoring System for Mice & Rats by Kent Scientific Corporation™), and body temperature was maintained between 35-38.5℃ through the use of the heating pads. Heart rate and SpO2 were measured using a pulse oximeter specifically designed for mice (MouseSTAT ® Jr. Rodent Pulse Oximeter). Pulse oximeter readings were taken from the left or right hind foot. If the SpO2 for a mouse dropped below 75%, then the mouse was placed on supplemental 100% oxygen at 0.8L/min provided through a nose cone. Respiratory rate was measured through the counting of thoracic excursions. Mucus membrane color was monitored through visual observation. PWRs were monitored using a reflex instrument that applied a consistent noxious stimulus of 300 g. A positive PWR was defined as any movement made by the mouse in response to the noxious stimulus. Physiologic monitoring was continued every 5 minutes through the duration of anesthesia until the return of the PWR or other spontaneous movement. If there was no sign of a return of the PWR or other spontaneous movement 90 minutes from the time a cocktail containing an α2-adrenergic agonist had been injected, the mouse was reversed using atipamezole (Revertidine ™ Modern Veterinary Therapeutics, 5 mg/mL) administered IP.
Once there was a return of the PWR or other spontaneous movement, the mice were monitored for the return of their RR. To evaluate the return of the RR, all mice were manually placed in dorsal recumbency. If the mouse was able to right itself by placing one or more paws back onto the surface they were lying on, they were determined to have regained their RR.
Results
Note: All mice in this study were put onto supplemental 100% oxygen at 0.8L/min while under a surgical plane of anesthesia since SpO2 consistently dropped below 75% within 15 minutes of the loss of the PWR.
Experiment 1: Telazol and Dexmedetomidine with and without Buprenorphine ER
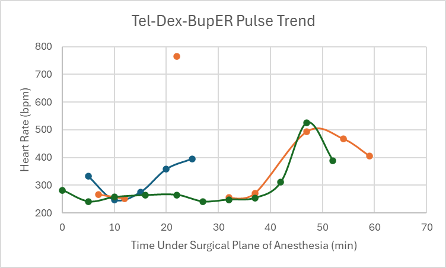
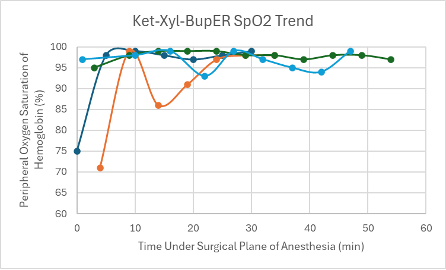
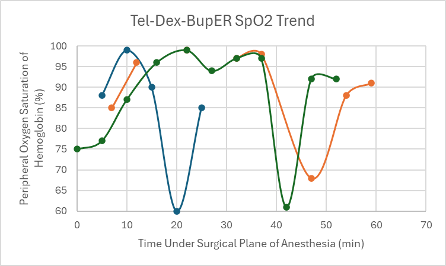
The first mouse that was given the Tel-Dex cocktail received a dose from the cocktail described in Table 1 at double the concentration. The dose this mouse received was 39 mg/kg Tel and 0.48 mg/kg Dex. A surgical plane of anesthesia was successfully achieved for 26 minutes in the male CD-1 mouse. However, it took the mouse 59 minutes to regain its RR after regaining its PWR. Within this time, it experienced a series of undesirable recovery symptoms, such as head tremors and circling. The mouse also experienced a distinct drop in SpO2 down to 60% (Figure 11), while on supplemental oxygen, which prompted its reversal with atipamezole administered through an IP injection at T = 24 minutes. Due to these undesirable side effects during anesthesia and recovery, the cocktail formulation was adjusted to that found in Table 1, and a new low dose was administered with the addition of BupER in female transgenic C57BL6 mice (n = 2). A surgical plane of anesthesia was successfully achieved in each mouse for 54 and 63 minutes. Again, there was a noticeable drop observed in their SpO2 down to 61% at T = 42 minutes and down to 68% at T = 47 minutes for each mouse, respectively, while on supplemental 100% oxygen (Figure 11). Due to these drops in SpO2, the mice were reversed within 10 minutes. The mouse that was under surgical anesthesia for 54 minutes successfully regained its RR 38 minutes after it started showing signs of spontaneous movement in its head. While recovering, it exhibited evidence of respiratory effort due to a persistent respiratory noise. The mouse that was under surgical anesthesia for 63 minutes never recovered its RR and passed away 52 minutes after regaining its PWR. A gross necropsy was performed, and no abnormal findings were made except for segments of mildly hyperemic small intestine. Because this cocktail combination presented a significant drop in SpO2 for all mice (n = 3), and negative recovery symptoms were consistently observed, no further mice were tested using this cocktail.
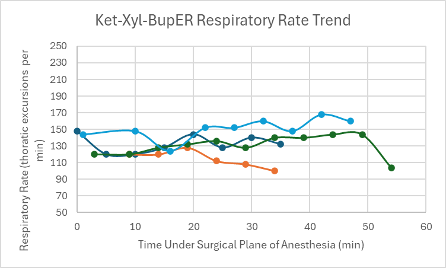
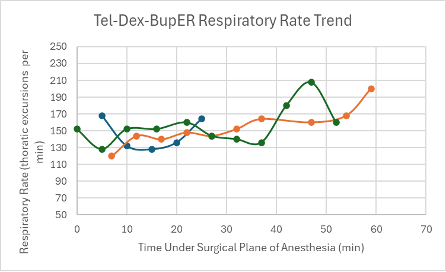
All three mice experienced a spike in heart rate at the same time that their SpO2 dropped. Apart from these spikes, the heart rate in each mouse was depressed, but trended upwards over the course of the anesthesia. The respiratory rates for the mice remained relatively stable and within normal parameters (80-200 breaths per min) and trended upwards towards the end of the duration of the surgical plane of anesthesia (Figure 19).
Experiment 2: Ketamine, Dexmedetomidine, and Acepromazine
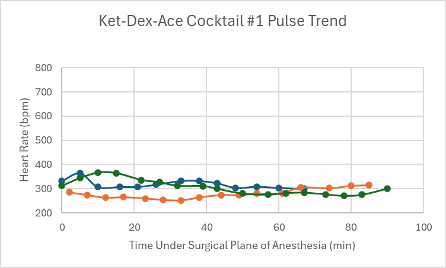
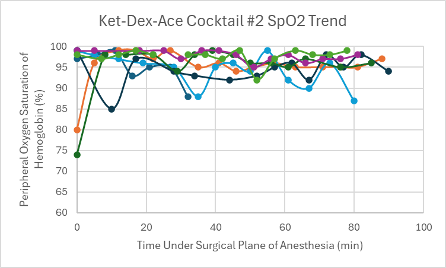
The first version of this cocktail (Ket-Dex-Ace Cocktail #1) was administered in male Agouti and female transgenic C57BL6 mice (n = 2 and n =3, respectively). The first male mouse received a low dose along with all three of the female mice, but only the female mice achieved a surgical plane of anesthesia. Despite never reaching a surgical plane of anesthesia, it took the first male mouse 66 minutes to regain his RR. For the females, the surgical plane of anesthesia lasted between 74-91 minutes, and they regained their RR within 11-15 minutes of regaining their PWR. Since the first male did not achieve a surgical plane of anesthesia at a low dose, the second male was given a middle dose of Ket-Dex-Ace #1. Again, a surgical plane of anesthesia was not achieved, but RR reflexes were not regained until after 69 minutes. Since the first version of the cocktail failed to achieve a surgical plane of anesthesia in both male Agouti mice, the remaining mice (n =6) were tested using the Ket-Dex-Ace Cocktail #2 found in Table 1.
A low dose was administered to one male CD-1 mouse and male and female transgenic C57BL6 mice (n = 3 and n = 2, respectively). The low dose of this version achieved a surgical plane of anesthesia that lasted between 36-98 minutes in all mice that received it (n = 5). Once the PWR reflex was regained, it took these mice an average of 26.6 minutes to regain their RR. The last mouse tested with this cocktail, a female transgenic C57BL6 mouse, received a mid-dose and achieved a surgical plane of anesthesia that lasted for 32 minutes. She regained her RR 18 minutes after regaining her PWR.
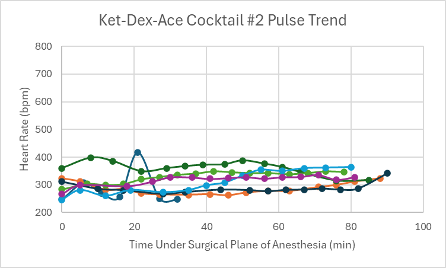
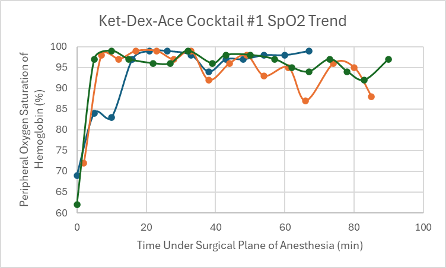
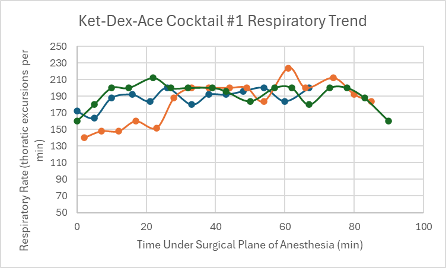
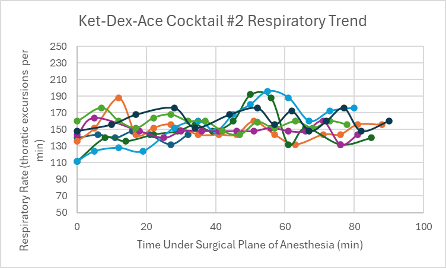
Both versions of this cocktail resulted in a consistent, slightly depressed heart rate with a slight trend upward towards the end of the duration of the surgical plane of anesthesia (Figures 4 and 5). Additionally, both cocktails showed variable SpO2 values with a slight trend downwards in the range of 85-99% despite all mice being put onto supplemental 100% oxygen early on in their surgical planes of anesthesia (Figures 12 and 13). The respiratory rates of the mice were slightly variable, but overall, were within normal parameters and consistent throughout the duration of the anesthesia (Figures 20 and 21).
Apart from vitals, the mice that received these cocktails were noticeably sedate and minimally ambulatory 32-88 minutes after fully regaining their RR.
Experiment 3: Alfaxalone and Dexmedetomidine with and without Buprenorphine ER
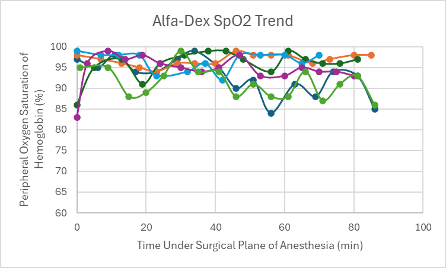
In this experiment, the Alfa-Dex cocktail was tested both with and without the addition of BupER. A low dose without the addition of BupER was administered in male and female Transgenic C57BL6 mice (n =3 and n = 2, respectively). It successfully achieved a surgical plane of anesthesia in all mice (n = 5) that lasted between 46-89 minutes. After regaining their PWR, it took the mice an average of 26 minutes to regain their RR.
A mid-dose without the addition of BupER was administered in one female transgenic C57BL6 mouse and successfully achieved a surgical plane of anesthesia that lasted for 91 minutes. Once the PWR was regained, it took this mouse another 37 minutes to regain its RR.
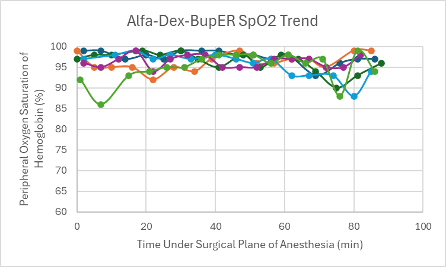
A low dose of this cocktail with the addition of BupER was administered in male and female transgenic C57BL6 mice (n = 2 and n = 2, respectively) and female Heterozygous Nude Nu/J mice (n = 2). A surgical plane of anesthesia was successfully reached in all of the mice (n = 6), lasting between 66-92 minutes. After regaining their PWR, it took the mice an average of 12.3 minutes to regain their RR.
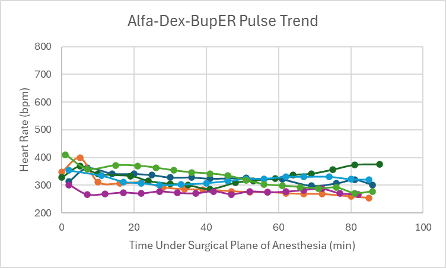
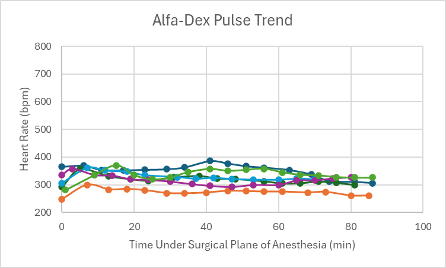
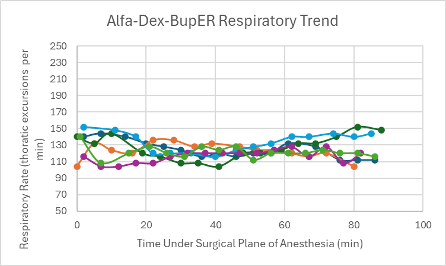
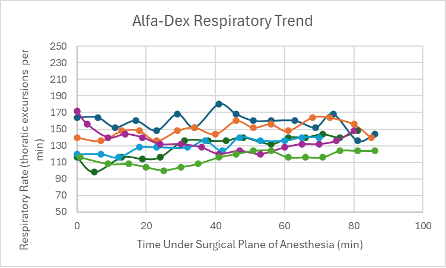
The heart rates for all mice that received this cocktail with and without BupER were consistent, though slightly depressed, throughout the duration of the surgical plane of anesthesia (Figures 6 and 7). The SpO2 was higher and more consistent for the mice that received this cocktail with the addition of BupER compared to the more variable SpO2 trends seen in the mice that received this cocktail without the addition of BupER (Figures 14 and 15). Similarly, the respiratory rates for the mice that received BupER were more consistent with each other than the respiratory rates of the mice that did not receive BupER (Figures 22 and 23). Nevertheless, both respiratory rate trends remained within normal limits.
Experiment 4: Alfaxalone and Midazolam with and without Buprenorphine ER
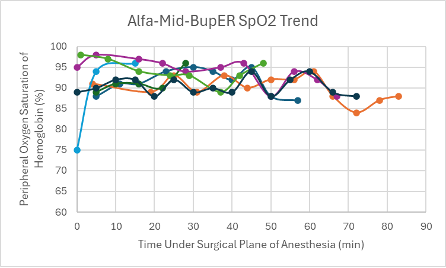
The first three trials done with the cocktail did not include BupER. A low dose was administered in female transgenic C57BL6 mice (n = 2), and a mid-dose was administered to a male transgenic C57BL6 mouse. A surgical plane of anesthesia was reached in both female mice, but only lasted 4-12 minutes, while a surgical plane of anesthesia was never reached in the male mouse. Despite only being under a surgical plane of anesthesia for a short period of time, it took the female mice 108-112 minutes to regain their RR after regaining their PWR. For the male mouse, it took 121 minutes for the RR to be regained.
The remaining trials done with this cocktail included BupER. To begin, a low dose was administered to a female Heterozygous Nude Nu/J mouse. A surgical plane of anesthesia was reached and lasted for 33 minutes. It took this mouse 34 minutes to regain its RR after regaining its PWR. Next, a mid-dose was administered to male and female transgenic C57BL6 mice (n = 1 and n =1, respectively). A surgical plane of anesthesia was achieved in both, but only lasted between 10-13 minutes. After regaining their PWR, it took the mice 65-81 minutes to regain their RR. To finish off the trials for this cocktail, a high dose was administered to the remaining mice (n = 6). A surgical plane of anesthesia was reached in all of the mice (n = 6) that lasted between 24-87 minutes. The average time it took to regain the RR after regaining the PWR was 36.7 minutes.
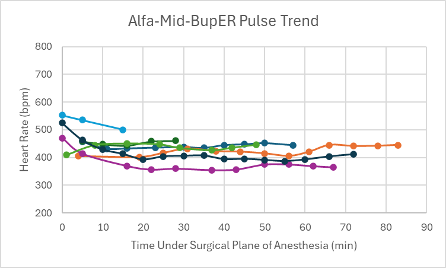
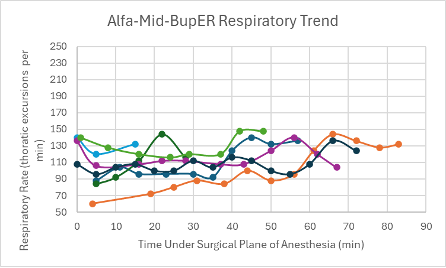
The heart rates for the mice that received this cocktail were noticeably higher and within normal parameters (350-700 bpm) in comparison to the heart rates of mice that received other cocktails (Figure 8). For the majority of mice that successfully reached a surgical plane of anesthesia, their SpO2 trended downward throughout the duration of the anesthesia (Figure 16). Respiratory rates were variable but overall trending upward throughout the duration of anesthesia (Figure 24).
Control A: Ketamine and Xylazine with Buprenorphine ER
All mice in this group received a low dose of the cocktail (n = 6). The mice that successfully achieved a surgical plane of anesthesia for 20 minutes or more included male CD-1, Agouti, and transgenic C57BL6 mice (n =1, n =1, and n =1, respectively) and a female transgenic C57BL6 mouse (n = 1). The surgical plane of anesthesia for these mice lasted between 35-55 minutes. On average, these mice regained their RR 13.3 minutes after regaining their PWR. Two mice, a male transgenic C57BL6 mouse and a male transgenic DBA mouse, achieved a surgical plane of anesthesia, but it only lasted between 2-5 minutes. After regaining their PWR, it took these mice 21-28 minutes to regain their RR.
For the mice that successfully achieved a surgical plane of anesthesia, their heart rates were slightly depressed, but were relatively consistent throughout the duration of the anesthesia (Figure 1). One mouse experienced a noticeable drop in SpO2 down to 86% at T = 14 minutes (Figure 9). After this drop, the SpO2 trended upwards for the remainder of the surgical plane of anesthesia. The respiratory rates for the mice in this control group were variable but within normal parameters (Figure 17).
Control B: Ketamine and Dexmedetomidine with Buprenorphine ER
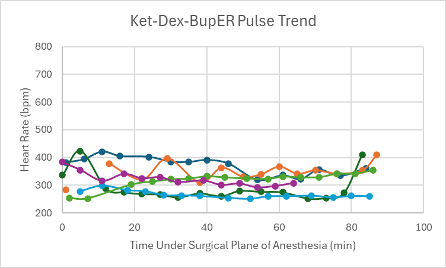
Mice in this group received either a low dose (n = 5), or a mid dose (n = 1). The mice that received a low dose included male and female transgenic C57BL6 mice (n = 3 and n = 1, respectively), and a female Heterozygous Nude Nu/J mouse. All of the mice given a low dose successfully achieved a surgical plane of anesthesia (n = 5) that lasted between 49-93 minutes. After regaining their PWR, it took these mice an average of 15.8 minutes to regain their RR. The mouse in this group, a male transgenic C57BL6 mouse that received a mid-dose, successfully achieved a surgical plane of anesthesia that lasted for 90 minutes. Two minutes after regaining its PWR, it regained its RR.
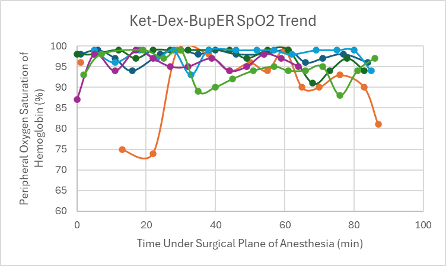
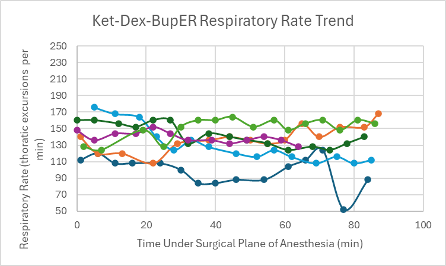
All mice that received this cocktail had a depressed heart rate that was relatively consistent throughout the duration of the surgical plane of anesthesia (Figure 2). The majority of mice also experienced a trend downward in SpO2 over the duration of the anesthesia (Figure 10). Respiratory rates were quite variable with an overall trend downwards over time (Figure 18).
Figure 2: Heart rate trend in mice under a surgical plane of anesthesia after the administration of the Ket-Dex-BupER cocktail.
Figure 3: Heart rate trend in mice under a surgical plane of anesthesia after the administration of the Tel-Dex-BupER cocktail.
Figure 4: Heart rate trend in mice under a surgical plane of anesthesia after the administration of the Ket-Dex-Ace Cocktail #1.
Figure 5: Heart rate trend in mice under a surgical plane of anesthesia after the administration of the Ket-Dex-Ace Cocktail #2.
Figure 6: Heart rate trend in mice under a surgical plane of anesthesia after the administration of the Alfa-Dex-BupER cocktail.
Figure 7: Heart rate trend in mice under a surgical plane of anesthesia after the administration of the Alfa-Dex cocktail.
Figure 8: Heart rate trend in mice under a surgical plane of anesthesia after the administration of the Alfa-Mid-BupER cocktail.
Figure 9: SpO2 trend in mice under a surgical plane of anesthesia after the administration of the Ket-Xyl-BupER cocktail.
Figure 10: SpO2 trend in mice under a surgical plane of anesthesia after the administration of the Ket-Dex-BupER cocktail.
Figure 11: SpO2 trend in mice under a surgical plane of anesthesia after the administration of the Tel-Dex-BupER cocktail.
Figure 12: SpO2 trend in mice under a surgical plane of anesthesia after the administration of the Ket-Dex-Ace Cocktail #1.
Figure 13: SpO2 trend in mice under a surgical plane of anesthesia after the administration of the Ket-Dex-Ace Cocktail #2.
Figure 14: SpO2 trend in mice under a surgical plane of anesthesia after the administration of the Alfa-Dex-BupER cocktail.
Figure 15: SpO2 trend in mice under a surgical plane of anesthesia after the administration of the Alfa-Dex cocktail.
Figure 16: SpO2 trend in mice under a surgical plane of anesthesia after the administration of the Alfa-Mid-BupER cocktail.
Figure 17: Respiratory trend in mice under a surgical plane of anesthesia after the administration of the Ket-Xyl-BupER cocktail.
Figure 18: Respiratory trend in mice under a surgical plane of anesthesia after the administration of the Ket-Dex-BupER cocktail.
Figure 19: Respiratory trend in mice under a surgical plane of anesthesia after the administration of the Tel-Dex-BupER cocktail.
Figure 20: Respiratory trend in mice under a surgical plane of anesthesia after the administration of the Ket-Dex-Ace Cocktail #1.
Figure 21: Respiratory trend in mice under a surgical plane of anesthesia after the administration of the Ket-Dex-Ace Cocktail #2.
Figure 22: Respiratory trend in mice under a surgical plane of anesthesia after the administration of the Alfa-Dex-BupER cocktail.
Figure 23: Respiratory trend in mice under a surgical plane of anesthesia after the administration of the Alfa-Dex cocktail.
Figure 24: Respiratory trend in mice under a surgical plane of anesthesia after the administration of the Alfa-Mid-BupER cocktail.
Discussion
Experiment 1: Telazol and Dexmedetomidine with and without Buprenorphine ER
Throughout the duration of the surgical plane of anesthesia, this cocktail had unstable SpO2 and heart rate trends. Due to the large drops in SpO2, each mouse likely experienced hypoxia, which caused a spike in heart rate to occur at the same time (Figure 3). In addition to these trends, all mice experienced adverse side effects as the anesthesia wore off, such as head tremors, circling, and death. For such reasons, we would not recommend this cocktail for mice in a laboratory setting unless more research was done into alternative doses that may prove to be more effective, reliable, and safe.
Experiment 2: Ketamine, Dexmedetomidine, and Acepromazine
The first version of this cocktail did not achieve a surgical plane of anesthesia for 20 minutes or more in two of the mice that received it. Both mice were male Agouti, so it is possible that these drugs are not as effective in this strain of mouse compared to other strains. The second version of this cocktail was more successful in producing a surgical plane of anesthesia for all of the mice that received it. During reformulation, the concentration of ketamine and acepromazine was reduced, while the concentration of dexmedetomidine was increased. Since dexmedetomidine is an α2-adrenergic agonist, increasing its concentration likely increased the analgesia produced in the mice, which could have contributed to the more successful surgical plane of anesthesia. That being said, both versions of this cocktail produced extensive sedation even after the righting reflex was regained, and the mice required comparably longer monitoring and heat supplement throughout recovery. Since this type of prolonged sedation did not occur in mice of the control cocktail group, Ket-Dex-BupER, it is likely that this effect can be attributed to the acepromazine in this cocktail. However, more research would need to be done to explain why this drug potentially causes prolonged sedation in mice. Though the reformulation of this cocktail was effective, we would recommend using it for longer procedures or surgeries and instead using alternative cocktails for more routine procedures.
Experiment 3: Alfaxalone and Dexmedetomidine with and without Buprenorphine ER
For this experimental group, an even number of mice received the cocktail with and without the addition of BupER. Those that did receive BupER showed signs of a noticeably more stable SpO2 and respiratory rate throughout the duration of the surgical plane of anesthesia. Interestingly, BupER, known to cause respiratory depression11 which is attributed to a worse SpO2, surprisingly seemed to aid in the stabilization of this vital sign. This study does not have a clear explanation for this phenomenon, but it could be beneficial to do further research into how BupER potentially improves the performance of alfaxalone and dexmedetomidine. For the purposes of this study, we would recommend using BupER in addition to this cocktail. Overall, the cocktail proved to have the most stable and consistent heart rate trend throughout the duration of the anesthesia, and no adverse side effects were observed, so we would recommend using it as an effective alternative injectable anesthetic for mice.
Experiment 4: Alfaxalone and Midazolam with and without Buprenorphine ER
Mice that received this cocktail had a noticeably higher heart rate compared to the other experimental and control cocktails. Out of all of the cocktails, this was the only cocktail that did not include an α2-adrenergic agonist, which is known to cause a decreased heart rate and respiratory rate12. Instead, this cocktail was made up of alfaxalone, a GABA agonist, and midazolam, a sedative and coinduction agent to alfaxalone. It could be due to these differing chemical properties that the mice that received this cocktail had higher heart rates. Despite having heart rate trends that were higher than other cocktails, this cocktail produced a relatively and consistently low SpO2 which is not desirable. In addition to this, it showed a variable respiratory rate that was on the lower end of normal parameters.
All of the mice that received this cocktail without the addition of BupER did not successfully reach a surgical plane of anesthesia for 20 minutes or longer, so we would recommend adding BupER when using this cocktail. However, even with BupER, two mice did not successfully stay in a surgical plane of anesthesia for 20 minutes, out of a total of nine mice. Though this cocktail is effective in the majority of mice with the addition of BupER, more research should be done to see if a more reliable version of the cocktail can be created.
Final Recommendations
Out of the two control cocktails used in this study, one showed more reliable results and more desirable vital signs throughout the duration of the surgical plane of anesthesia. This cocktail was Ket-Dex-BupER. The dose we would recommend using for this cocktail for the best results is 50 mg/kg of Ketamine with 0.60 mg/kg of Dexmedetomidine.
Out of the four experimental cocktails evaluated in this study, two showed the most potential in effectively providing a surgical plane of anesthesia for at least 20 minutes. These cocktails were Alfa-Dex-BupER and Ket-Dex-Ace. The dose we recommend for the Alfa-Dex-BupER cocktail is 50 mg/kg of Alfaxalone and 0.50 mg/kg of Dexmedetomidine with the addition of 1.0 mg/kg of Buprenorphine ER. The dose we recommend for the Ket-Dex-Ace cocktail is 50 mg/kg of Ketamine, 0.60 mg/kg of Dexmedetomidine, and 0.50 mg/kg of Acepromazine.
References
- Jiron JM, Mendieta Calle JL, Castillo EJ, Abraham AM, Messer JG, Malphurs WL, Malinowski C, Grove K, Reznikov LR, Zubcevic J, Aguirre JI. Comparison of isoflurane, ketamine-dexmedetomidine, and ketamine-xylazine for general anesthesia during oral procedures in rice rats (oryzomys palustris). J Am Assoc Lab Anim Sci. 2019 Jan 1;58(1):40-49. doi: 10.30802/AALAS-JAALAS-18-000032.
- Buitrago S, Martin TE, Tetens-Woodring J, Belicha-Villanueva A, Wilding GE. Safety and efficacy of various combinations of injectable anesthetics in BALB/c mice. J Am Assoc Lab Anim Sci. 2008 Jan;47(1):11-7.
- Alexander RS, Canver BR, Sue KL, Morford KL. Xylazine and overdoses: trends, concerns, and recommendations. Am J Public Health. 2022 Aug;112(8):1212-1216. doi: 10.2105/AJPH.2022.306881.
- Hagan L, David EM, Horton AR, Marx JO. Effects of midazolam/dexmedetomidine with buprenorphine or extended-release buprenorphine anesthesia in C57BL/6 mice. J Am Assoc Lab Anim Sci. 2024 Mar 1;63(2):172-181. doi: 10.30802/AALAS-JAALAS-23-000063.
- Navarro KL, Huss M, Smith JC, Sharp P, Marx JO, Pacharinsak C. 2021. Mouse anesthesia: the art and science. ILAR J. 62(1–2):238–273. doi: 10.1093/ilar/ilab016.
- Lin HC, Thurmon JC, Benson GJ, Tranquilli WJ. 1993. Telazol – a review of its pharmacology and use in veterinary medicine. J Vet Pharmacol Ther. 16(4):383–418. doi: 10.1111/j.1365-2885.1993.tb00206.x.
- Erickson RL, Blevins CE, Souza Dyer C, Marx JO. 2019. Alfaxalone-xylazine anesthesia in laboratory mice (mus musculus). J Am Assoc Lab Anim Sci. 58(1):30–39. doi: 10.30802/AALAS-JAALAS-18-000010.
- Young MS, Kelly JC, Anderson SR, Riffle LA, Spears SL, Kalen JD, Suess-Radford E, Gulani J. 2024. Subcutaneous alfaxalone-xylazine-buprenorphine for surgical anesthesia and echocardiographic evaluation of mice (mus musculus). J Am Assoc Lab Anim Sci. 63(1):49–56. doi: 10.30802/AALAS-JAALAS-23-000090.
- Murdock MA, Riccó Pereira CH, Aarnes TK, Cremer J, Lerche P, Bednarski RM. Sedative and cardiorespiratory effects of intramuscular administration of alfaxalone and butorphanol combined with acepromazine, midazolam, or dexmedetomidine in dogs. Am J Vet Res. 2020 Jan;81(1):65-76. doi: 10.2460/ajvr.81.1.65.
- Animal Husbandry Information – Animal Resource – University of Rochester Medical Center. Rochester.edu. Published 2021. Accessed November 25, 2024. https://www.urmc.rochester.edu/animal-resource/animal-husbandry.aspx
- Dahan A, van Lemmen M, Jansen S, Simons P, van der Schrier R. Buprenorphine: a treatment and cause of opioid-induced respiratory depression. Br J Anaesth. 2022 Mar;128(3):402-404. doi: 10.1016/j.bja.2021.12.001.
- Meyer R, Fish R. Anesthesia and analgesia in laboratory animals (second edition): chapter 2 – pharmacology of injectable anesthetics, sedatives, and tranquilizers. American College of Laboratory Animal Medicine; 2008:27-82. doi: 10.1016/B978-012373898-1.50006-1
About the Author
Eleanor Harrison is an undergraduate microbiology student on the pre-veterinary medicine track at the University of Rochester. With an interest in pharmacology, she was thrilled to have this opportunity to better understand the world of veterinary anesthetic drugs through this independent research project. She is also fascinated by viruses and would be interested in pursuing future research involving zoonotic viruses and their emergence in humans and other animal species. Apart from these interests, Eleanor has a deep passion for conservation, and hopes to pursue a veterinary career working with wildlife and exotic animals through conservation efforts.
Cite this Article
Harrison, E. and LeMoine, D. (2025). Surgical Anesthetic Potential of Injectable Cocktails Containing Acepromazine, Alfaxalone, Dexmedetomidine, Ketamine, Midazolam, Telazol, and Xylazine with and without the Addition of BuprenorphineER in Mice. University of Rochester, Journal of Undergraduate Research, 23(2).
JUR | Creative Commons Attribution 4.0 BY International License