
Why don’t bats get cancer?
Rochester scientists discover that long-lived bats resist cancer thanks to strong immune systems and protective genes—findings that could offer new insights into human aging and disease prevention.

Bats offer clues to treating COVID-19
Bats carry many viruses, including the one behind COVID-19, without becoming ill. University of Rochester biologists are studying the immune system of bats to find potential ways to “mimic” that system in humans.

2019 d.health Summit addresses social determinants of health
Now in its fifth year, the d.health Summit will address the non-medical factors the affect health, such as economic security, education, physical environment, employment, social support networks, and access to care.

‘Selfish’ genetics amplify inflammation, age-related diseases
Research from Rochester biologists shows that a class of genomic parasites may cause more harm than previously thought, triggering inflammation that causes age-related diseases.

Why do naked mole rats live long, cancer-free lives?
Rochester biologists were surprised to see that despite its remarkable longevity, the naked mole rat still has cells that undergo senescence, like the cells in much shorter-lived mice.

d.health Summit showcases innovative approaches to aging
Nearly one-fifth of the U.S. population will be 65 or older by the year 2030. To address this challenge, the University’s third annual d.health Summit will convene thought leaders across health care, finance, technology, and policy.
Study identifies key factor in DNA damage associated with aging
There are many examples of DNA damage being associated with aging, but never has a reduction in DNA damage been shown to extend lifespan. Rochester research has made this connection, and identified an enzyme that can be targeted to reduce that damage.

d.health Summit 2017 focuses on the future of aging
Underpinning this year’s theme are a selection of topics that discuss the future of medicine, technology, and healthcare policy as they relate to aging Americans. This national forum will be held at The New York Academy of Sciences, at the World Trade Center in New York City.
Protein found that initiates DNA repair
Researchers who specialize in the study of aging have identified a protein that may serve as a first responder, activating a “longevity gene” known as sirtuin 6 and setting in motion a cascade of molecular first responders to repair damaged DNA.
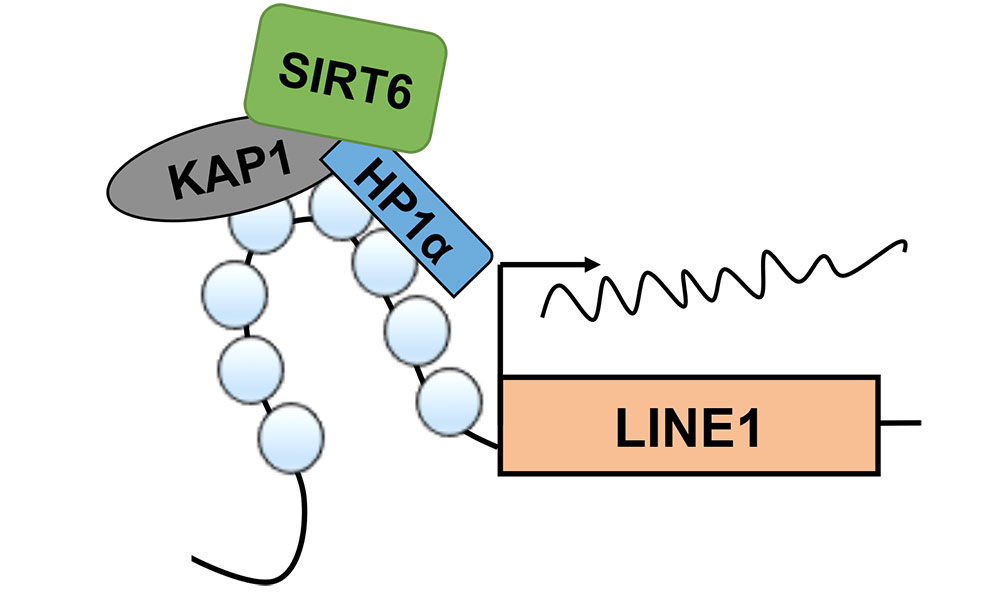
Parasitic DNA stops “jumping” when protein takes charge
Biology researchers Vera Gorbunova and Andrei Seluanov report that the “jumping genes” in mice become active as the mice age when a multi-function protein stops keeping them in check in order to take on another role. A protein called Sirt6 is needed to keep the jumping genes—technically known as retrotransposons—inactive.
