The brain’s system for removing waste is driven primarily by the pulsations of adjoining arteries, University of Rochester neuroscientists and mechanical engineers report in a new study. They also show that changes in the pulsations caused by high blood pressure slow the removal of waste, reducing its efficiency.
This might explain the association between high blood pressure and Alzheimer’ disease, the researchers say. Alzheimer’s, the most common cause of dementia among older adults, is characterized by abnormal clumps and tangled bundles of fibers in the brain.
The study, reported in Nature Communications, builds upon groundbreaking discoveries about the brain’s waste removal system by Maiken Nedergaard, co-director of the University’s Center for Translational Neuromedicine. Nedergaard and her colleagues were the first to describe how cerebrospinal fluid is pumped into brain tissue and flushes away waste. Subsequent research by her team has shown that this glymphatic waste removal system is more active while we sleep and can be damaged by stroke and trauma.
This latest research shows “in much greater depth and much greater precision than before” how the glymphatic system functions in the perivascular spaces that surround arteries in the outer brain membrane, says Douglas Kelley, an assistant professor of mechanical engineering and an expert in fluid dynamics. His lab is collaborating with Nedergaard’s team as part of a $3.2 million National Institute on Aging grant.
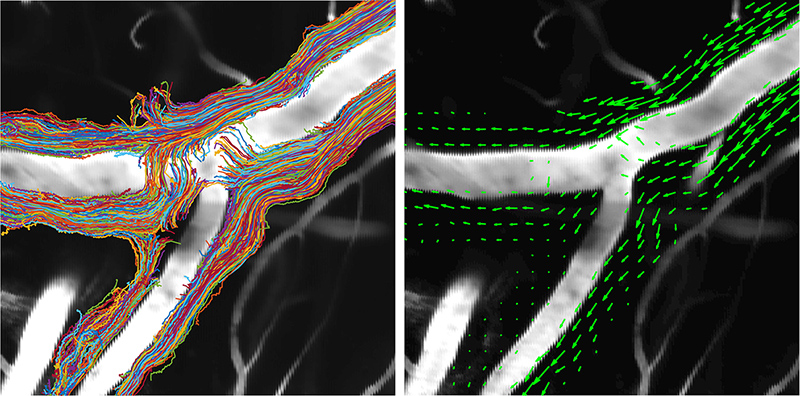
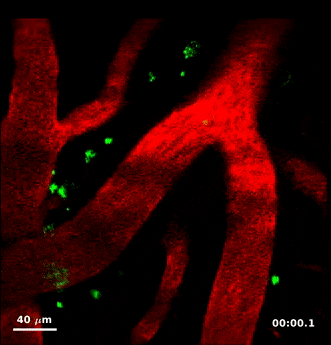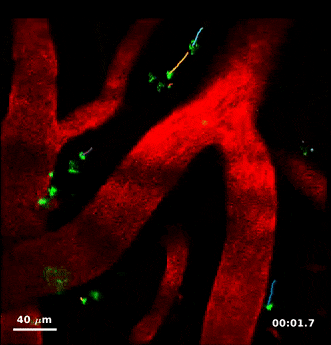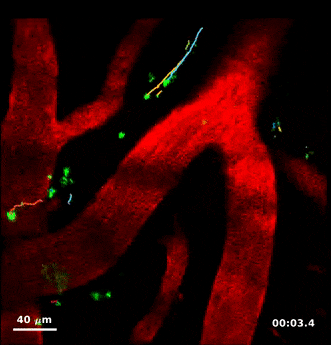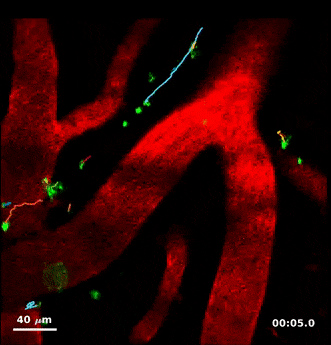
For this study, Humberto Mestre, a PhD student in Nedergaard’s lab, injected minute particles in the cerebrospinal fluid of mice, and then used two-photon microscopy to create videos showing the particles as they moved through the perivascular spaces.
Jeff Tithof, a postdoctoral researcher working with Kelley, then used an automated particle tracking code developed by the Kelley lab that can follow a dot from frame to frame of a video, and after a few frames estimate its velocity and acceleration. From a typical video spanning “tens of minutes,” the code could track more than 20,000 individual particles and derive about a million total measurements to quantify their movements, which show up as “comet tails” behind each of the tracked dots in the videos.
By analyzing videos from experiments involving multiple mice, “we were able to gather a really rich data set of what’s actually happening with this flow,” Kelley says.
The researchers found that:
- The flow of cerebrospinal fluid is synchronized with the heartbeat, as seen in the animation below. “One of the main points of this paper is that this flow is mostly – and maybe purely — driven by the flexing of the artery wall,” Kelley says.
- When high blood pressure was induced in the mice, the flow slowed down and was not as efficient. Artery walls are muscular; they have to flex harder in order to hold the same shape when there’s more pressure inside, and they get stiffer,” Kelley says. “And that changes the waveform of the flexing of the artery wall.”
“That’s highly interesting because early onset high blood pressure is known to be a risk factor for Alzheimer’s in humans,” Kelley says. “There are still a few steps between what we did and chronic high blood pressure. But it may be that high blood pressure—by reducing the pumping of cerebrospinal fluid so that waste isn’t cleared out as well — is one mechanism that leads to Alzheimer’s.”
The findings should resolve a debate among researchers about whether the flow of cerebrospinal fluid in the glymphatic system is in the same direction as arterial blood flow, as demonstrated by this paper, is opposite, or is random molecular diffusion, Kelley says. “This is going to quell a lot of those debates.”
The collaboration with Nedergaard also includes Department of Mechanical Engineering faculty members John Thomas (a coauthor of this paper) and Jessica Shang, along with Ali Ertürk with the Institute for Stroke and Dementia Research at Ludwig Maximilians University of Munich. Other coauthors of this paper include Ting Du, Wei Song, Weiguo Peng, Amanda Sweeney, and Genaro Olveda of Nedergaard’s lab.
Learn more about Rochester’s role in understanding the brain’s waste removal system.
Maiken Nedergaard was recently awarded the 2018 Eric K. Fernstrom Foundation Grand Nordic Prize for her work that led to the discovery of the brain’s unique waste removal system and its role in a number of neurological disorders. Nedergaard maintains labs at the Medical Center and the University of Copenhagen.
In 2012, Nedergaard’s lab was the first to reveal the brain’s unique process of removing waste, dubbed the glymphatic system, which consists of a plumbing system that piggybacks on the brain’s blood vessels and pumps cerebral spinal fluid (CSF) through the brain’s tissue, flushing away waste.
Nedergaard’s lab has since gone on to show that the glymphatic system:
- works primarily while we sleep,
- could be a key player in diseases like Alzheimer’s,
- is disrupted after traumatic brain injury,
- may be enhanced by moderate alcohol consumption, and
- could be harnessed as a new way to deliver drugs to the brain.




