Overview
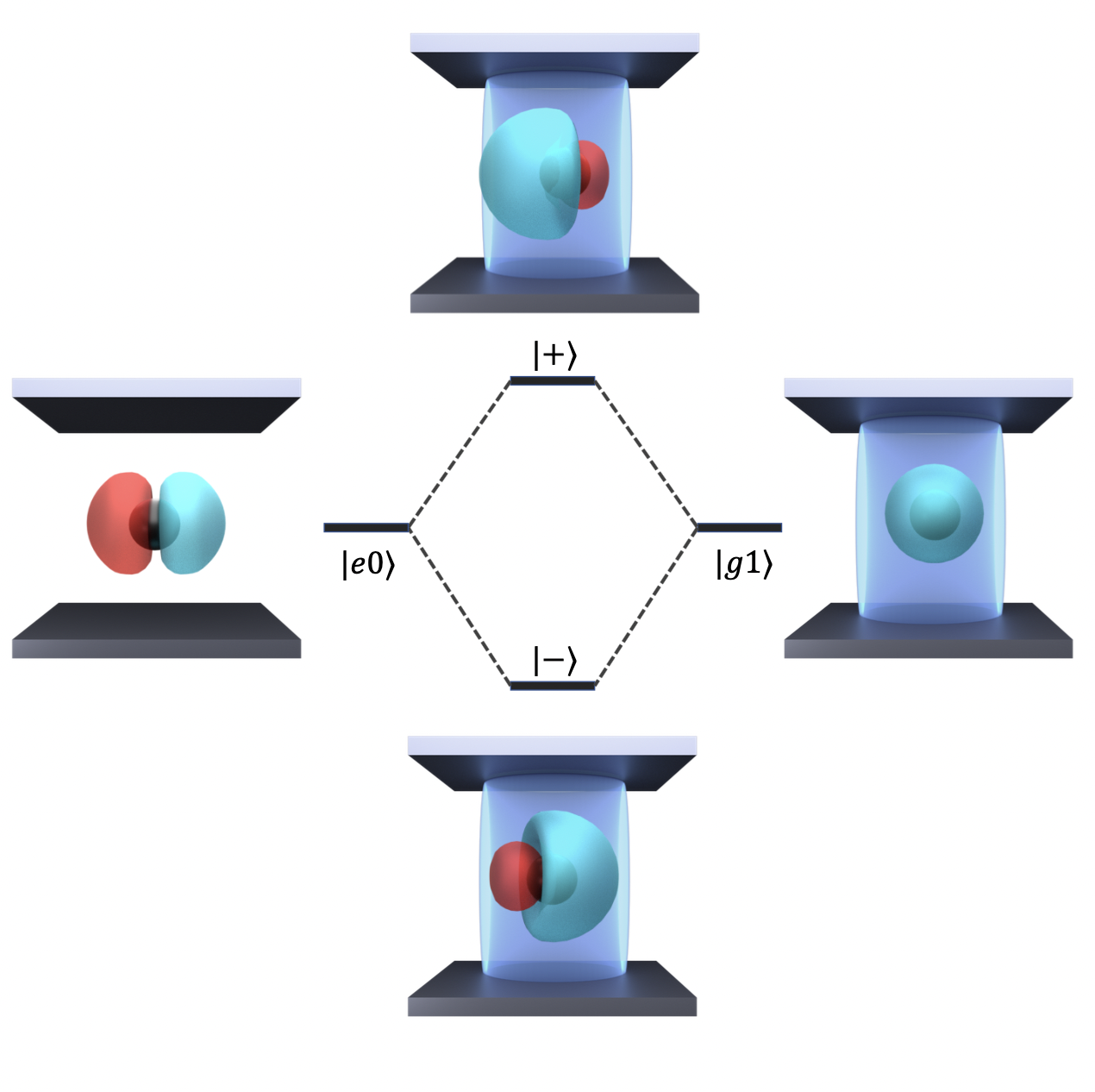
Coupling molecules to a quantized radiation field inside an optical cavity creates a set of photon-matter hybrid states, so called polaritons. These polariton states hybridize the curvatures from both the ground and the excited electronic states and have shown a great promise to alter the photochemistry of molecules. Unlike traditional photochemistry, which uses light as an energy source, polariton chemistry uses quantized photons as active chemical catalysts to significantly change the shape of the potential energy surface in molecular systems, and thus, open up new possibilities to tune and control chemical reactions.
A useful way to think of the polariton state is a hybridized orbital consisting of the ground electronic state of the molecule hybridized with the excited electronic state. For example, for a simple hydrogen atom, the electronic state component of the polariton would be a hybridized 1S-2P state, which is not possible for an isolated atom in the absence of the cavity. Alternatively, hybridizing the excitonic or vibronic states of a molecule with the photonic states of a cavity forms exciton- or vibrational-polartions, respectively, which are commonly referred to as molecular polaritons.
